|
Thus, Sulphur will share six of its valence electrons with all the fluorine atoms that result in forming six single bonds between S and F. As every atom follows the octet rule to attain a stable structure, the Fluorine atom will share one valence electron of the Sulphur atom. So place it in the centre and all Fluorine atoms around it like this:įluorine atom needs only one valence electron to complete its octet. Sulphur atom will take the central position as it is less electronegative than Fluorine. This structure helps us know the bond formations in the molecule and the arrangement of electrons in it. The Lewis Dot structure of any molecule is a pictorial representation of the atoms involved in forming the structure and its individual valence electrons. Thus SF6 has 48 valence electrons that will help us draw the Lewis Dot Structure of SF6. Total number of valence electrons in SF6 – Valence electrons of Sulfur + Valence electrons of Fluorineįluorine has seven valence electrons, but as there are six Fluorine atoms in this molecule, we will multiply this number by 6. Here we will find out the total number of valence electrons for SF6 by adding the valence electrons for both Sulfur and Fluorine atoms. 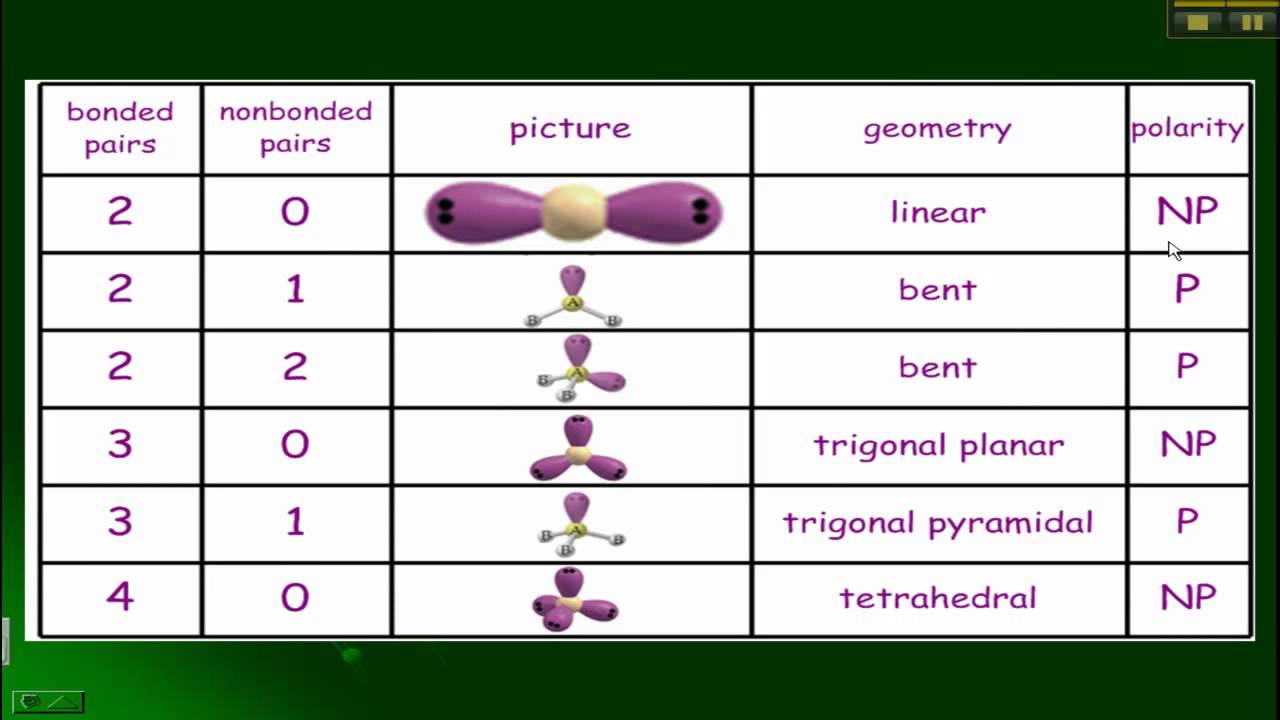
To determine the Lewis Structure of any molecule, we first need to know the total number of valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
